3D molecular model of thalidomide
Online discussions on the merits of alternative medicine can get quite heated. And its proponents, given enough time, will inevitably cite the same drug as “evidence” of the failings of science. Call it Gavura’s Law, with apologies to Mike Godwin:
As an online discussion on the effectiveness of alternative medicine grows longer, the probability that thalidomide will be cited approaches one.
A recent comment on my own blog, regarding the homeopathic product Traumeel, is typical:
If the scientific method is all that separates an accepted claim, ie Thalidomide, Vioxx, Bextra, Darvon, from mere anecdote, of what benefit is the Science?
As a non-scientist consumer, I’ll take the anecdotes and my own experience. Thank you.
If scientists want to be taken seriously, they must stop selling themselves to the highest bidder becoming corporate whores without a shred of decency. To my mind, that’s how the claims for Thalidomide, Vioxx, Bextra, Darvon were accepted, making the scientific method utterly worthless.
To this commenter, “science has been wrong before.” And that invalidates science, and apparently validates homeopathy. It’s a fallacious argument. But does thalidomide actually represent a failing of science-based medicine? No, not even close. It’s so wrong, it’s not even wrong. Thalidomide is good example of the importance of science-based medicine and why allowing alternative medicine to be sold in the absence of good science is a concern.
The broad strokes of thalidomide causing thousands of birth defects are well known. But the details are important to understand the implications to regulation, and to science-based medicine, today.
Thalidomide was first marketed in 1958 in Germany by Chemie Grünenthal. While its developmental origins are somewhat unclear, it was initially marketed as a treatment for seizures, and later as an anti-nauseant and sedative. At the time, barbiturates were frequently used as sedatives. And compared to barbiturates, which were highly toxic in overdose, thalidomide was well tolerated, even in overdose. Based on its apparent safety, no prescription was required. Eventually its attractiveness as an anti-nauseant led to its use in pregnancy for morning sickness.

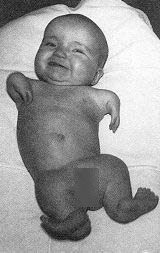
It wasn’t known at the time, but fetal exposure to thalidomide between days 35 and 48 was causing severe limb and organ defects in 20-30% of children. In the 1950’s it wasn’t even recognized that drugs could cross the placenta and cause adverse effects to the fetus. Thalidomide hadn’t been tested on pregnant animals prior to marketing for use in pregnancy. Regulators didn’t require it. And the consequences were horrific.
Thalidomide became a popular drug because of its apparent safety and effectiveness, and it was marketed alone and in combination with other drugs in Germany, the UK, Canada, and other countries. But within a few years, babies started being born with characteristic limb and organ deformities. In 1961, two independent researchers identified thalidomide as the likely causal agent. The manufacturer sought to undermine and discredit the findings, but it was clear – the drug had caused catastrophic harm to thousands of fetuses. Subsequent animal testing confirmed this.
The USA largely escaped the thalidomide tragedy – due to stronger regulations, and the action of a single employee.
American drug regulations have evolved over time. The Pure Food and Drugs Act (1906) did not require that drugs be tested for safety and efficacy before sale. It was the regulator’s responsibility to demonstrate a product was unsafe. In 1938, the S.E. Massengill company manufactured a liquid version antibiotic sulfanilamide, mixing it with diethylene glycol -a poison more commonly used as antifreeze. When patients started dying, a massive recall was implemented – but not before over 100 people died. The tragedy brought about significant reforms to drug regulations in the form of the Food, Drugs and Cosmetics Act (1938) that required manufacturers to obtain FDA approval for any drug sold, prior to any sale.
Frances Oldham Kelsey was a reviewer with the FDA in 1960, and was responsible for evaluating the thalidomide marketing application. Kelsey refused to approve the drug in the absence of safety data. Her concern was peripheral neuropathy – not teratogenicity, which wasn’t even considered at the time. She described the application process in a paper she published in 1965:
The New Drug Application for thalidomide was presented in September, 1960. The drug had been marketed in Germany since 1957 where it was available without prescription, and in Great Britain since 1958. However, it was felt that the evidence submitted in the application was not adequate to indicate the safety of the drug. In particular, although this drug appeared to be remarkably nontoxic in animals and human beings, little or no information was available concerning its absorption, distribution in the body, or its excretion. Since the possibility existed that the low toxicity of the drug in certain species might be related to poor absorption in those species, and that under certain conditions the absorption in other species might be increased, further work was requested relative to the metabolism of the drug.
As the FDA studied the side effect profile, the teratogenicity of the drug became clear as and the application for licensure was discontinued. Kelsey was hailed as hero. Yet despite the refusal to formally approve the product for sale, millions of doses had been administered as part of “clinical trials” (in name only). Yet due to Kelsey’s refusal to approve the drug, the drug was not widely used, and only 17 children were born in the USA with thaldomide-induced effects.
Regulators worldwide acted to ensure another thalidomide tragedy would not occur. In 1962 the Kefauver-Harris Drug Amendments were passed, increasing the rigor of the drug approval process, requiring the demonstration of safety and effectiveness via objectively designed and executed clinical trials. These amendments also implemented the requirement for adequate preclinical trials before any human studies, including animal studies to evaluate fetal risks. Marketing requirements were also implemented, restricting manufacturers from making unfounded, unsubstantiated claims of safety and efficacy. Finally, the amendments ushered in the requirement for manufacturers to collect and report all adverse events associated with drug use – measures designed to capture any events not otherwise identified in pre-marketing clinical trials. Similar actions occurred in other countries. Today, over 100 countries now collaborate with the World Health Organization to pool adverse event reporting and look for signals of drug harms.
Lessons Learned
The worldwide disaster of thalidomide led to most of the current framework we have in place today to evaluate the safety and efficacy of drug products. Ironically, these are the same requirements that manufacturers of supplements and other “alternative” medicine products have been largely successful in circumventing. In the USA, it’s DSHEA, which (as blogged about regularly at SBM) removed the onus of demonstrating safety and efficacy from the manufacturer and put the requirement to demonstrate harm on the FDA – exactly the same scenario as drugs in the early 1900’s. In Canada, the Natural Health Product regulations has introduced a lowered bar for non-drug supplements: Today even homeopathic remedies are deemed safe and effective and approved with unique recommended uses.
The Resurrection of Thalidomide
Almost 30 years after thalidomide was withdrawn, it’s back on the market in the United States, Canada and many other countries. It’s been found to be effective for a number of conditions including erythema nodosum leprosum, multiple myeloma, and is being investigated for efficacy in an array of other conditions. There’s an analog of thalidomide now marketed, lenalidomide, also used to treat cancer. To minimize the teratogenicity risk, intensive programs are in place to minimize any possibility of use in pregnancy. So while the drug has known harms, it seems to be highly effective for some medical conditions where few effective alternatives exist. Access, therefore, is based on a careful evaluation of the risks and benefits.
Does Thalidomide Invalidate Science-Based Medicine?
Cases like thalidomide provide a good example of why SBM authors argue against regulatory double-standards, and advocate for a single, science-based standard for evaluating all products: drugs as well as supplements. There is no intrinsic reason to think any product, regardless of its source, is safe or effective – we must evaluate it, objectively. Thalidomide’s history is a cautionary tale reminding us that assumptions without good evidence can lead to terrible consequences.
And that leads to the logical fallacies that the thalidomide retort represents. Thalidomide is an effective drug for some conditions – but it is a teratogen. It’s not alone in this regard – other drugs have been identified as teratogens. The sale of any drug is based on an evaluation of the known benefits and risks.And getting back to the comment I received at the top, those that support alternative medicine will reject the science-based approach: This is the system that gave us thalidomide (and Vioxx, etc). Therefore, it’s a failure, and we should reject it. This is the perfect solution fallacy.
Admittedly, medicine is not perfect. Regulators don’t identify all the harms before a drug is licensed. Drug manufacturers can behave badly. And drugs don’t always work the way we want them to, and they can cause harms. They commenter I cite above took objection to an evaluation of homeopathy. Yes, homeopathy has no toxic side effects (usually) and causes no teratogenicity – but it also has no demonstrated efficacy beyond placebo effects. The harms and problems of science-based approaches add no support to the efficacy claims of any alternative medicine system. Homeopathy, acupuncture, and reiki don’t become effective because drugs can have side effects. SBM may not be perfect, but it delivers the goods.
More generally, comparing science-based medicine to any alternative medical system a is false dichotomy. Unlike the different alternative medicine systems (homeopathy, naturopathy, reiki, etc) which are typically based around a fixed set of rules, the only thing that science-based medicine interventions have is common is that they work. SBM is not immutable to change – treatment shown to be safe and effective become medicine, and those that are not, are discarded. So although thalidomide causes birth defects, it is also an effective medication when used properly. We don’t have to make a choice between a system that produced drugs that cause birth defects, and alternative medicine. We choose to use treatments where the expected benefits outweigh the known risks.
Citing thalidomide as an argument against science-based approaches is also a straw man argument. The way drugs are regulated today bears little resemblance today to when thalidomide was licensed and sold. Safety standards are far more rigorous, in part because of the lesson of thalidomide. Today, the only products that are not subject to these same strict safety and efficacy standards are usually the alternative medicine treatments.
Citing thalidomide is also an appeal to fear. We hear the word and we immediately think of children born with birth defects. We don’t want that to happen. But like the perfect solution fallacy, the catastrophic side effects of thalidomide in pregnancy add no merit to to claims of efficacy of any alternative medicine treatment or system. We still need the evidence.
Conclusion
Thalidomide was a very real tragedy with a huge human cost. It’s most important lesson is that assumptions of safety and efficacy, in the absence of evidence, can be catastrophic. Citing thalidomide as a a reason to reject science-based medicine is not only fallacious, it reflects a fundamental lack of understanding of the lessons learned.